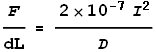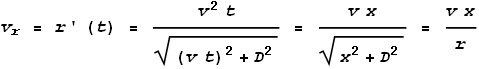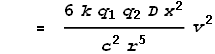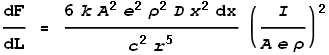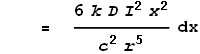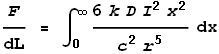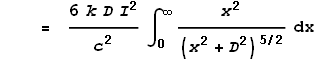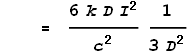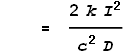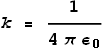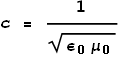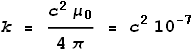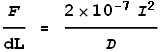Magnetism – Proof of Concept
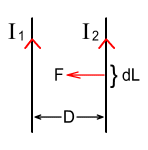
Force between wiresTo test the theory out, we’ll look at the magnetic force between parallel wires. Suppose we have two parallel wires as shown below:
The wires carry currents of I1 and I2 respectively and are separated by a distance D. What will be the magnetic force F experienced by each wire per unit length dL be? Magnetic force equations tell us that:
To simplify the calculations, let’s say the current in both wires is I. The force per unit length will be:
And this is the result we are aiming for.
Using the VDCLNow let’s see what we predict for magnetic force using the VDCL.
First we note that that each wire holds equal numbers of protons and electrons, giving the
wires a neutral charge. Most of those electrons are fixed while others are ‘free
floating’ and able to carry a current. We can eliminate the fixed electrons and a
corresponding number of protons from our consideration because they have no net effect.
What remains then is only the floating (current carrying) electrons and an equal number of
unmovable protons.
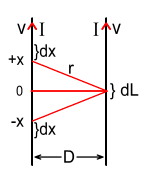
Let’s look at the force produced in a small section of the right-hand
wire of length dL, by two small sections of the left wire of length dx
located at distances of +x and –x from the centre.
The force on the wire segment dL is made up of the repulsive force from
the electrons and the attractive force from the protons. Assume each wire has the same
cross sectional area of A and the velocity of the electrons in each wire
is v. Also let the charge located within a dx segment be
q1, and the charge located within the dL
segment be q2.
Where r is the distance from dL to dx and is given by:
Now to calculate the force from the protons. To do this we must consider the velocity of the protons relative to the electrons. The velocity vr at point x relative to dL can be determined from the rate of change of the distance r.
Using this velocity function in the VDCL, the (stronger) force from the protons at -x can be given by:
The (weaker) force from the protons at +x can be given by:
The total force can be calculated by adding forces from both protons and electrons at both positions (+x and -x). Doing this we get:
This shows the force in terms of the velocity v. But we want it in terms of the current I. To do this we use the electron density conversion formula:
Where ρ is the electron density per cubic volume, A is the cross sectional area of the wire and e is an electron’s charge. We also need to express the charges q1 and q2 in similar terms:
Substituting these values of velocity and charge into the force expression we get:
This gives the force from just the two points at dx. To determine the full force from the entire length of the left wire we must integrate the above expression over all positive values of x:
Coulombs constant k can also be expressed as:
Maxwell’s and Ampere’s laws tell us that:
Extracting ε0 and substituting it into the equation for k we see that:
Substituting this value of k into the force equation we get:
This is the exact result we were after! Namely, the VDCL has computed the magnetic force equation.
ConclusionBased on the above calculations, we can see that the VDCL can be used to
determine the exact result as Maxwell’s magnetic force equations.
Wait... a mistake?The above calculation only determines the force from the protons in the
Left wire upon the electrons in the Right wire. But the Right wire also has protons that
will be attracted to the electrons of the Left. If this is included the total force per
unit length will be twice as large. Are our results too small by a factor of 2?
|