Atomic Nuclei
Looking for cluesWe know little about a neutron’s structure. It is impossible to see
anything that tiny using present technology. Therefore we must look for clues that can be
combined with our understanding of electrical forces in order to piece together a picture
of the nucleus.
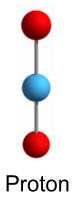
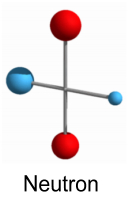
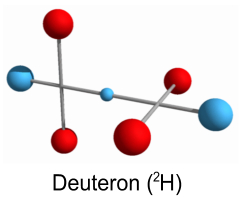
Higher order nucleiWe’ll start by reviewing the models we have thus far for a proton, neutron and deuteron (a ‘heavy hydrogen’ nucleus: 2H). These models appear stable and represent the simplest forms of the
hydrogen nucleus and its components. How would we proceed in building the nucleus of
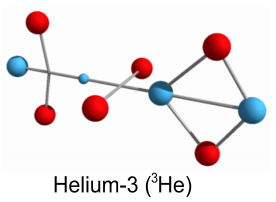
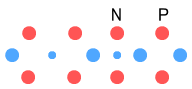
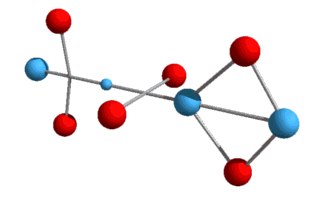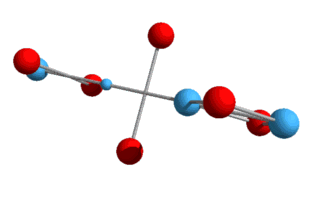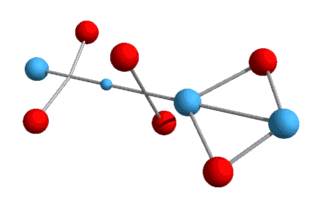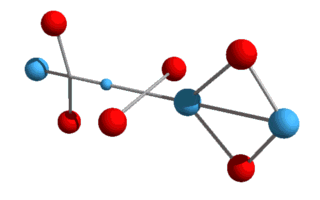
higher elements, such as helium, lithium, etc., and various isotopes? What we now have is a Helium-3 nucleus, consisting of two protons and a neutron. Helium-3 is known to be a stable isotope of Helium, and from a visual perspective at least, it does appear stable. Just as with deuterium, this Helium-3 structure will need to rotate in order to hold the up-quarks at an altitude. We can also draw it in a flattened two-dimensional perspective:
Here the up-quarks are shown in the same plane, although they really
alternate at right angles.[Note: particles are not drawn to scale in either 2D or 3D
models.]
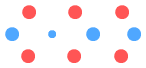
There are a large number of up-quarks close together here, unlike the
Helium-3 which spread them apart. Notice that when deuterium was converted to Helium-3,
the up-quarks bent away from the centre, making it difficult to add further protons. Thus
we would expect Lithium-4 to decay into Helium-3 by rejecting that end proton, which is
what happens (proton emission).
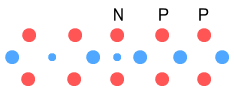
This effectively converts the right-hand proton into a neutron, making the isotope Hydrogen-3. We know that Hydrogen-3 is largely stable with a half-life of 12 years and that it decays into Helium-3 by ejecting an electron (beta decay). According to the above diagram, all this does seem likely. The right-hand proton is wide enough to accommodate an electron. Let’s then try adding another neutron:
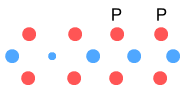
This will make Hydrogen-4. Unlike Hydrogen-3, Hydrogen-4 is very unstable.
It has a half life of 10-22 seconds and decays by emitting neutrons. In the
above diagram we note that the neutron is being stretched horizontally due to an excess
number of negatively charged particles (electrons and down-quarks) close together.
Therefore it is quite likely that this additional neutron will be rejected as a whole.
Adding additional neutrons will make Hydrogen-5,6 and 7, all of which are unstable and
decay by ejecting those neutrons.
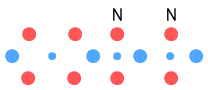
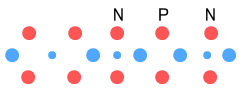
This gives us Helium-4, which is of course very stable. The diagram
appears to confirm this. The protons are spaced by a neutron and that partly unstable
Hydrogen-3 electron is now better secured by the additional up-quarks.
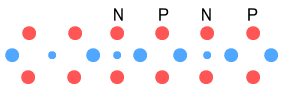
Both Lithium-5 and Helium-5 are known to be unstable, and the diagram
appears to confirm this. For Lithium-5 there are too many protons (or more specifically,
up-quarks) close-together and too few negative charges to secure them; hence there would
be much repulsion. As might be expected, Lithium-5 decays via proton emission into
Helium-4.
This gives us Lithium-6, which is stable.
Nuclear Magnetic ResonanceI could go on in this manner but you get the basic idea. By adding protons
and neutrons to the end of the chain we can make different elements and isotopes. Certain
isotopes will be stable and others not. Stability will depend on how close protons get to
each other and how compressed the electrons and down-quarks are. For the unstable isotopes
we can also predict what sort of decay mechanisms might occur.
It is easy to see how these mini ‘bar magnets’ can align themselves with an external magnetic field. Nucleons (protons and neutrons) arranged in a ball could also align themselves but in the process they would repel each other; similar to how it is difficult to align two nearby bar magnets in the same direction.
Large nucleiFor small elements the number of neutrons and protons are roughly
equivalent but as the elements become heavier we notice a steady increase in the number of
neutrons versus protons. The reason suggested for this is that extra neutrons help minimise
repulsion between protons. If we look at the chain model, this is most likely true. But
here the neutrons do more than act as ‘spacer washers’ between protons; they
also provide electrons upon which the protons can secure themselves.
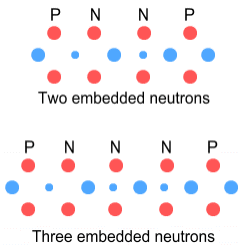
As can be seen, when there are two neutrons it appears the protons can attach to their outer negative charges (down-quarks and electrons) thus holding them stable. When there are three neutrons, the middle neutron is difficult to secure and there is also a large number of negative charges close together. Hence the three-neutron situation is unlikely to be stable.
RulesCertain rules seem to govern the formation of stable isotopes.
Helium-3 seems to violate rule 1. However its right-most added proton is
not necessarily attaching to another proton because the two nucleons of deuterium are
symmetrical and either could be considered a proton or neutron. Hydrogen-3 violates rule
2, as it is mostly stable. This stability is possible due to the Helium-3 proton widening
itself sufficiently for the electron to be inserted.
MysteriesThe preceding observations don’t fully explain all isotopes. For
example Helium-8 is more stable than Helium-7, and Lithium-11 more so than Lithium-10. In
both cases the larger isotope has more neutrons and we would expect less stability. In the
case of lithium, an interesting study [2] found the size of Lithium-11
and Lithium-12 nuclei was much greater than Lithium-10. It’s possible that additional
neutrons have somehow separated from the rest of the chain and are floating at some
distance. For example Lithium-11 may be made of a Lithium-9 and two neutrons held at some
distance. How this could happen is unclear. |